1. General Information
Welcome to the School of Medical Sciences
Welcome to your Postgraduate Taught Programme in the School of Medical Sciences within the Faculty of Biology, Medicine and Health at the University of Manchester. The University has a worldwide reputation based on high quality teaching and research, and I am sure that your programme will provide a solid foundation for your future career success.
Within the School and the wider Faculty, our goal is to create an environment that allows you to excel and reach your full potential. Offering access to first-class facilities and strong links with regional health-service providers, our postgraduate programmes are designed to meet the diverse needs of all our students. The curriculum of our taught programmes provides the knowledge and skills you will need in your subject area and all our Masters programmes include an opportunity to carry out an independent research project on topics spanning all areas of biomedical research from molecular to experimental biology and clinical medicine. While subject areas cover a broad range, all our taught programmes have two common aims:
- To develop your skills in your chosen field of study
- To enhance your knowledge within the field you have chosen. Whether you are a graduate, professional or have a clinical background, the programmes have been tailored to meet your specific needs.
As a student of the School of Medical Sciences, you will be expected to take responsibility for your degree, within a supportive environment that fosters your development and helps prepare you for your future career. This handbook will be a useful resource as you progress through your programme. It provides programme-specific information that I am sure that you will find helpful throughout your study. If however, you have questions or would like some further advice, please do not hesitate to contact the people listed in this handbook for further information and assistance.
I wish you every success as you embark upon your programme, and in your future career.
Dr Carol Yates
Director of Postgraduate Taught Education
School of Medical Sciences
Faculty of Biology, Medicine and Health
Updated version
Academic year 2019/20
Welcome to the MSc in Advanced Therapy Medicinal Products
Advanced Therapy Medicinal Products (ATMPs) are medicines for human use that are based on genes, cells or tissue, sometimes referred to as “living drugs”. ATMPs are a fast paced and rapidly evolving area of medicine which are increasingly being used to treat a broad range of malignant and non-malignant diseases offering ground breaking new opportunities for the treatment of disease and injury.
The UK government has recognised that there is an opportunity for the UK to become a beacon of activity for ATMPs. In 2018 three National Advanced Therapy Treatment Centres (ATTCs) were funded by Innovate UK to form a National ATTC Network. The purpose of this network is to support and foster collaborative working across the NHS and industry to accelerate patient access to advanced therapies. Central to this goal is the provision of a comprehensive and robust education and training programme to train the workforce and researchers of the future.
The MSc in Advanced Therapy Medicinal Products is an integral part of the strategic development of Advanced Therapies in Greater Manchester and more broadly across the UK. The course is designed to equip the researchers, clinicians and health professionals of tomorrow with a broad understanding of the challenges of bringing an ATMP through development and to patients. This unique multi-disciplinary master’s degree will utilise a range of expertise and knowledge from expert clinicians, preclinical development scientists as well as industry and regulatory experts to deliver an exciting and unique programme catering for health care professionals and scientists.
The objective of the course is to enable students to undergo comprehensive training in all aspects of the field of ATMP development and delivery. In this unique course design students can choose to follow laboratory (wet-lab) based or clinical (dry-lab) research-based content with optional modules available covering a wide variety of ATMP indications. This will allow students to diversify depending on their own interests and backgrounds. Three principle methods will be used to achieve this:
- Taught units: Four compulsory taught units will provide the theoretical framework to understand Advanced Therapy Medicinal Products
- Dissertation: Two dissertation projects will enable students to design, deliver and interpret projects related to Advanced Therapy Medicinal Products:
- For wet-lab students this will comprise one 30 credit project which may be undertaken outside of a lab environment and one 90 credit project which will be undertaken within a lab environment and supported by a laboratory base supervisor.
- For dry-lab students this will comprise two 60 credit projects with allocation of a suitable supervisor related to the chosen project topic.
- Experience: As students on the ATMP MSc, you will have the opportunity to spend time with a clinical or laboratory based team working in the area of advanced therapies. This will give you the opportunity to work alongside a skilled workforce designing and contributing to advanced therapy clinical studies.
As programme director for the MSc in Advanced Therapy Medicinal Products, I would like to formally welcome you onto the programme, and also take the opportunity to invite you to contact me should any concerns or clarifications arise during your graduate learning experience.
Points of Contact
| Programme Directors | Michelle Davies The National Institute for Health Research Clinical Research Facility Christie Hospital NHS Foundation Trust Wilmslow Road Manchester M20 4BX Tel: 0161 918 7875 michelle.davies@christie.nhs.uk Professor Robert Wynn |
| Programme Administrator | TBC The University of Manchester Room 1.485 Stopford Building Oxford Road Manchester M139PT Tel: 0161 275 ATMP@manchester.ac.uk |
| ATMP Student Representative | To be appointed by students post registration. The student rep will be nominated from the selection of the students who would like to volunteer for the position. The student rep is required to feedback to the programme directors and administrator on any issues or queries that the students have. They will be required to attend official programme committees throughout the year. |
| eLearning Support | You can contact eLearning to receive support at elearning@manchester.ac.uk. However, you must enter “FBMH eLearning – MSc in Advanced Therapy Medicinal Products” in your email subject header. This will help ensure your request reaches us quickly. Further information on BMH eLearning can be found here. |
| General IT Support | Contact the Service Desk via the online Knowledge Base http://servicedesk.manchester.ac.uk/portal/ss and report any IT failure or submit a service request. Call the Service Desk on: 0161 306 5544 (or ext 65544). Opening hours are: Monday to Friday 9am to 5pm. |
2. Overview of Programme
This programme offers both part-time and full time pathways. The structure of the programme is as follows:
PG Cert Advanced Therapy Medicinal Products
Complete the 4 course units indicated below. These units together contribute 60 credits to the programme.
| Course Unit | Title | Credits |
| Unit 1: MEDN 69910 | Research Methods | 15 credits |
| Unit 2: MEDN 69740 | Introduction to Advanced Therapy Medicinal Products | 15 Credits |
| Unit 3a: MEDN 69750 | Dry Lab pathway: Designing and delivering an ATMP clinical trial -manufacturing and regulatory complexities | 15 Credits |
| OR | ||
| Unit 3b: MEDN 66111 | Wet Lab pathway: Lab skills | 15 Credits |
| Unit 4 | One optional unit from list | 15 Credits |
MSc – 1 Year Full Time
Aims and Learning Outcomes
This ATMP master’s degree aims to deliver a broad and comprehensive overview with in -depth insights into the rapidly developing and cutting-edge field of Advanced therapies.
Students will undergo comprehensive training in all aspects of the field of ATMP development and delivery. In this unique course design students can choose to follow laboratory (wet-lab) based or clinical research-based content with optional modules available covering a wide variety of ATMP indications. This will allow students to diversify depending on their own interests and backgrounds. Our MSc in ATMPs is designed to equip the researchers, clinicians and health professionals of tomorrow with the necessary understanding of the challenges of bringing an ATMP through development and to patients. This will be achieved not only through the multi-disciplinary nature of the course content but through the range of expertise of the delivery staff. The programme is delivered by preclinical development scientists, researchers, regulatory professionals, clinicians and specialist experts currently working in ATMP development and delivery.
Intended Learning Outcomes of the Programme
On completion of the MSc you will:
- Clearly communicate complex concepts across the whole ATMP development pathway including but not limited to ATMP manufacturing techniques and supple chain issues, ATMP pre-clinical and clinical research design and ATMP clinical management; to a broad range of audiences within and across scientific disciplines and to non-scientific audiences
- Develop an ATMP clinical research protocol or lab based research project to inform the experimental direction of ATMP development.
- Evaluate ATMP research project data in the context of relevant, current literature, critiquing your own and/or published data
- Present research findings using a range of mediums including abstract development, an extended written report, delivering a poster presentation or delivering an oral defence.
Progression through the Programme
The table below shows progression through the programme. Students will be required to attend the allocated teaching days and to undertake the associated assessments. Progression from the PG Cert and the PG Dip will be dependent on successful completion of 60 credits, at the required standard. Similarly, progression from PG Dip to the MSc will depend on successful completion of 120 credits, at Masters Level.
| Units of Learning | Credits | Exit Award |
| 1. Taught Unit 1: Research Methods MEDN 69910 | 15 | |
| 2. Taught Unit 2: Introduction to Advanced Therapy Medicinal products MEDN 69740 | 15 | |
|
3. Taught Unit 3: DRY LAB Designing and delivering an ATMP clinical trial –manufacturing and regulatory complexities MEDN69750 OR 3. Taught Unit 3: WET LAB: Lab skills MEDN 66111 |
15 | |
| 4. Taught Unit 4: Optional module from list (see course unit summary doc) | 15 | PG Cert in Advanced Therapy Medicinal products (60 credit exit award for all pathways) |
|
5a. Research Project 1 – Dry Lab OR 5b. Research Project 2 – Wet Lab |
60 | PG Diploma in Advanced Therapy Medicinal products (120 credit exit award) |
|
6a. Research Project 2 – Dry Lab Or 6b. Research Project 2– Wet Lab |
60
90 |
MSc Advanced Therapy Medicinal products (180 credits – comprising Taught Units [60 credits] and Research Projects 1 & 2 [120 credits]) |
Course Units
Unit 1: Research Methods MEDN 69910 (15 credits)
Course Administrator: christie.finegan@manchester.ac.uk
The Research Methods Course is a 15 credit, interactive blended learning course which provides students with an introduction to key material required for the design, execution and interpretation of medical, scientific and clinically-related research and the production of a high quality dissertation.
The unit will run online over one semester. There will be online material opened on each topic at specific timetabled slots throughout the semester and face-to-face consolidation sessions.
The unit incorporates online material and 10 face-to-face sessions covering:
- Introduction to Blackboard and research methods online
- Dissertations skills – covering literature search, principles of academic writing, critical appraisal of publications, plagiarism and abstract writing
- Study design – covering project planning, time management and an overview of specific research designs, ethical issues and principles of good governance that apply to all clinical research
- Statistics – covering a basic introduction to statistical methods
- Communication skills – presentation skills and posters.
Aims
The unit aims to:
- Introduce students to the skills and knowledge to critically design, effectively implement,, ethically conducts and knowledgeably interpret research in biomedical and Human Sciences
- Provide students with life-long critical appraisal skills that they will be able to apply to any research evidence that comes before them.
Learning Outcomes
Om completion of this unit successful students will be able to:
| Knowledge and understanding |
|
| Intellectual skills |
|
| Practical skills |
|
| Transferable skills and personal qualities |
|
Teaching and Learning Methods
The course unit will be delivered predominantly through e-learning over one semester, with seven timetabled face-to-face sessions run by the Graduate Training Team and three face-to-face sessions run by the Library Training Team. A variety of online material will be utilised including web-based reading, podcasts, online discussions, tutor feedback, interactive exercises, self-assessment through MCQs and self-reflection.
The timetabled sessions will be a mix of tutor presentations, group discussions and workshops.
Assessment Methods
To help students appropriately focus their efforts throughout the unit, a combination of formative and summative assessments is offered.
All students must complete all formative assessments by the end of the semester.
Summative assessments will contribute a percentage towards the final mark. All students must complete three summative assessments in total. Two summative assessments “Critical appraisal of literature” and “Ethical issues related to clinical research” are compulsory for all programmes. Meanwhile, each programme of study can choose the third summative assessment from three optional assessments: Abstract writing, Grammar quiz, or Statistics quiz.
Feedback Methods
Students will be provided with personalised feedback for their summative assignments, within 15 working days.
Unit 2: Introduction to Advanced Therapy Medicinal Products (ATMP) MEDN 69740 (15 Credits)
Course unit lead: michelle.davies@christie.nhs.uk
Course administrator: TBC
Advanced Therapy Medicinal Products (ATMPS) offer promise as treatments for previously untreatable and high-burden diseases and significant investment has been made globally in order to continue to drive forward the fast paced development of these exciting new treatments. Unlike traditional medicines, ATMP development has unique considerations and complexities at every stage in the development process from pre-clinical, through trial development to clinical delivery. This module with give you the insight and understanding across all of these complexities and current developments in the field allowing you to become familiar with the key requirements of ATMP development from bench to bedside.
Module Content
This module will provide a comprehensive and broad overview of the development of advance therapy medicinal products from bench to bedside. You will begin by gaining an understanding of the molecular biology and broad science that underpins ATMP development and treatment. You will develop an understanding of the preclinical requirements of ATMP development to translate from bench to clinic including an overview of animal models commonly used to mimic human disease. You will understand the complexities involved in the manufacturing process of ATMPs including an understanding of commonly used viral and non-viral vectors and vector production. Additionally you explore the breadth of ATMP products currently under development and commercially available and how these can be used to modify disease states. You will explore and understand the range of pathological conditions which commonly utilise ATMPs for disease modification or control such as genetic disorders, cancer, rare and long term diseases. Additionally you will gain an understanding of the complexities and challenges of clinical delivery and management of an ATMP. This will include an overview of the associated toxicities of commonly used ATMP products and consideration of issues relating to long term follow up. Finally you will develop an understanding of the unique requirements required for the operational delivery of ATMPs including the need for specialist governance arrangements; this will include an overview of the various ATMP governing bodies and an understanding of the complexities of the ATMP regulatory landscape.
Learning Outcomes
- Demonstrate a critical and in depth understanding of the principles of ATMP development and clinical delivery and their application to disease.
- Critically evaluate and interpret the evidence presented in pivotal studies and seminal literature to identify current challenges related to ATMP clinical development.
- Communicate scientific information convincingly with a focus on ATMP development by developing a scientific abstract and presenting finding in a poster or oral presentation format.
- Apply understanding of the indications and clinical management of an ATMP for a specified disease to identify significant and relevant clinical issues and generate a novel ideas for their management, demonstrating consideration of the use of an interdisciplinary approach.
- Work in a group to assign a common solution to a complex problem relating to an unmet clinical need.
- Synthesise and develop rational arguments to contribute to key areas of controversy and discussion in the ATMP regulatory landscape.
- Provide and receive constructive feedback to and from peers and respond appropriately
Teaching and Learning Methods
Teaching on this course will be delivered by expert specialists and supported by academic staff from the University of Manchester and clinical experts allocated from an appropriate workplace/academic background. Some PhD students and post-doctoral research fellows will contribute to teaching where appropriate. In addition, opportunities for team-based learning will be provided.
This module offers the opportunity to engage with sessions led by internal and external experts. Learning will be integrally linked to course materials, group discussion and an interactive workshop. There will be opportunities for independent and group work. The course contains elements of self-assessment and peer and lecturer feedback.
Assessment Methods
Authentic assessments will be used during this module with appropriately placed formative assessments and early formative feedback. Assessment covers a number of communication types:
- Preparation of a scientific abstract in relation to one area of ATMP development (250 words)
- Prepare an oral or poster presentation which presents the work prepared in the previously presented abstract (10 min presentation and 5 min of questions)
- Critically appraise a paper with a focus on clinical delivery or regulatory challenges
A summary of the assessment structure in relation to learning outcomes and assessment weighting is presented in the table below:
| Formative Assessments | Learning Outcomes | Summative Assessments and Weighting | Learning Outcomes |
| Abstract practice | 1 + 3 | Abstract (20%) | 1, 2 +3 |
| Workshop to critically assess workflow issues in preclinical development with presentation at end | 1, 2 + 7 | Poster Presentation (50%) | 1, 2 + 3 |
| Journal club: choice of papers in relation to ATMP clinical indication and/or management for students work to review and critically present key findings | 4, 5 + 7 | Critical appraisal of a paper (30%) | 2, 4, 5 + 6 |
| Group Presentation of a regulatory issue | 6 + 7 | Formative only |
Feedback Methods
Students will be provided with personalised feedback for their summative assignments, within 15 working days.
Unit 3a: Designing and delivering an ATMP clinical trial – manufacturing and regulatory complexities MEDN 69750 (15 credits)
Course unit lead: mohammed.zubari@manchester.ac.uk
Course administrator: TBC
Advanced Therapy Medicinal Products (ATMPS) offer promise as treatments for previously untreatable and high-burden diseases and significant investment has been made globally in order to continue to drive forward the fast paced development of these exciting new treatments. This represents a paradigm shift in the treatment of many diseases which is likely to continue. The complexities of developing and delivering these treatments in turn require complex and novel approaches to ATMP clinical trial design. Furthermore the complex regulatory frameworks which are integral to ATMP trial governance and management are equally challenging. This module will provide a comprehensive and hands on approach to understanding ATMP clinical trial design and the related regulatory and governance frameworks.
Module content
This module will enable you to develop an understanding of the broad ATMP landscape, pathways and key stakeholders, reflecting on commercial and non-commercial trials to enable successful navigation of this complex environment. You will understanding the GMP requirements and functional practices involved in the manufacturing of ATMP treatments, use of raw materials (excipients) and associated tasks including assay development and the need for new governance structures to review ATMP treatments. You will understand the importance and relevance of the Good Manufacturing Practice (GMP) regulatory framework including specials. You will gain an understanding of Good Clinical Practice (GCP) regulatory requirements and functional practices involved in the design and development of ATMP treatments in clinical trials, This will encompass; roles and functions; need for risk assessment, Standard Operating Procedures (SOPs) and the ethical approval (HRA or Special) process. Furthermore you will undertake an exploration of the complex relationships, requirements and expectations of the MHRA, HTA and other organisations involved in the ATMP regulatory landscape. This will provide insight into the requirements needed to take an ATMP clinical trial from protocol design through regulatory approval in order to bring the newest ATMPs treatments to patients as quickly as possible.
Learning Outcomes
- Analyse the relationships between the appropriate ATMP governing bodies to demonstrate a critical understanding of the complexities of the ATMP regulatory landscape and apply this in relation to developing an ATMP clinical trial protocol.
- Synthesis and discuss the ATMP specific legislation in relation to GCP and GMP in the context of applying for regulatory approvals of an ATMP clinical trial.
- Process complex scientific information to present in lay terms. This will be demonstrated through the development of a clinical trial patient information sheet which must be written in plain language and formatted as per ethical and regulatory approval processes.
- Evaluate and apply all aspects of an ATMP clinical trial design including: (Developing a hypothesis, critical literature review, understanding of application of GCP and GMP, statistical information, study methodologies, endpoints and objectives, ethical considerations and legal and governance considerations ) using a clinical trial template.
Teaching and Learning Methods
Teaching on this course will be delivered by expert specialists and supported by academic staff from the University of Manchester and clinical experts allocated from an appropriate workplace/academic background. Some PhD students and post-doctoral research fellows will contribute to teaching where appropriate. In addition, opportunities for team-based learning will be provided.
This module offers the opportunity to engage with sessions led by internal and external experts. Learning will be integrally linked to course materials, group discussion and an interactive workshop. There will be opportunities for independent and group work. The course contains elements of self-assessment and peer and lecturer feedback.
Assessment Methods
Authentic assessments will be used during this module with appropriately placed formative assessments and early formative feedback. Assessment covers a number of communication types:
Formative:
- Introduction to protocol design and development setting up for end of module assessment
- Group work to develop experience of reviewing documents; developing insight and increasing experience in essential trial documentation
- Regulatory exam practice
Summative:
- Preparation of a clinical trial protocol (70%)
- Written exam on regulatory requirements for ATMPs (MCQs)
- Design and write an ATMP clinical trial patient information sheet for regulatory approval (30%)
Unit 3b: Lab Skills MEDN 66111 (15 credits)
Course uunit lead: susan.h.taylor@manchester.ac.uk
Course administrator: christie.finegan@manchester.ac.uk
The Laboratory skills unit aims to equip students with the theoretical understanding and practical skills relating to laboratory-based biomedical techniques to enable them to undertake, interpret and accurately record experimental research in the biomedical sciences.
The unit will consist of an induction session and three of the following workshops, providing theoretical or hands-on experience in the wet or dry laboratory environment:
| Induction (online) | Basic laboratory guidance, including health and safety regulations, keeping a laboratory notebook |
| Workshop 1 | Scientific Report writing |
| Workshop 2 | Manual handling in a laboratory, including dilutions and solution preparation |
| Workshop 3 | Microscopy: Theory of light, fluorescent and confocal microscopy; practical experience of light microscopy, including set-up and maintenance. |
| Workshop 4 | Staining techniques: histological staining, immunohistochemistry |
| Workshop 5 | Nucleic acid techniques 1: NA Extraction, NA amplification, electrophoresis |
| Workshop 6 | Nucleic acid techniques 2: Real-time PCR |
| Workshop 7 | Protein handling techniques1: Protein extraction/purification and analysis, including SDS-PAGE |
| Workshop 8 | Protein handling techniques 2: Western blot, Mass spectrometry/MALDI-TOF |
All students will complete the online induction and then take a further three workshops to complete the course unit, with no more than two from workshops 1-3.
Aims
The unit aims to equip students with the theoretical understanding and practical skills relating to laboratory-based biomedical techniques to enable them to undertake, interpret and accurately record experimental research in the biomedical sciences.
Learning Outcomes
On completion of this unit successful students will be able to:
| Knowledge and understanding |
|
| Intellectual skills |
|
| Practical skills |
|
| Transferable skills and personal qualities |
|
Teaching and Learning Methods
The teaching and learning processes will take the form of lectures, practical laboratory classes, laboratory demonstrations and eLearning (completion of online formative assessments)
Assessment Methods
| Workshop | Assessment Task | Length | Weighting Within Unit (if relevant) |
| Workshop 1 | Each workshop will be assessed by an online MCQ/SAQ | MCQ/SAQ exam | 20% |
| Workshops 2-8 | Each of the workshops will be assessed by a written practical report containing: (a) theoretical overview of the technique (b) report and interpretation of results obtained |
1000 words | 80% |
Feedback Methods
Students will be provided with personalised feedback for their summative assignments, within 15 working days.
Unit 4: Choice of Optional Units from Below
(a) Respiratory, Dermatology, Hearing Health and Musculoskeletal Pathways – Designing a translational medicine strategy for a clinical intervention MEDN 69632 (15 credits)
The purpose of this unit is to provide a framework to consider how to optimise the chances of success in the early clinical testing of a novel intervention. Despite having a potentially excellent drug, testing it in the wrong disease type, or at the wrong schedule or dose; or with the wrong combination of agent- could terminate an otherwise promising drug. Students will gain a foundation for considering which pre-clinical experiments can be done to inform on the optimal options for clinical testing. With the help of a tutor, students will choose a scenario to the practice of relevant translational medicine within the drug development environment and constructively critique it.
Aims
This unit aims to equip students with the knowledge and skills to:
- Understand the principles of designing translational medicine strategies for a clinical intervention including application of statistical theory
- Gain comprehensive knowledge of the translational research cycle
- Understand how pre-clinical experiments should be conducted to inform the design of a first in human clinical trial
- Prioritise the most pertinent pieces of pre-clinical data to inform the design of a first in human clinical trial
- Explore baseline and outcome measures that define success of an intervention
- Prioritise in a resource constrained environment which pre-clinical experiments are critical to attaining the next milestone and enlightening the clinical plan beyond that milestone
- Understand how to interpret data from Phase 1 clinical studies including measures of safety and efficacy
Learning Outcomes
| Knowledge and understanding |
|
| Intellectual skills |
|
| Practical skills |
|
| Transferable skills and personal qualities |
|
Teaching and Learning Methods
This unit will use Project-Based Learning and will be delivered in a blended format combining independent directed and undirected reading, one-to-one tutorials and discussions to explore examples and encourage candidates to draw upon their own clinical experience and reading.
Assessment Methods
Online academic critical reflection on clinical testing of a novel drug – 2,000 words (100%)
Feedback Methods
Students will be provided with personalised feedback for their summative assignments, within 15 working days.
(b) Molecular Pathology of Non-Malignant Disease MEDN 67520 (15 credits)
This unit will provide a broad based knowledge of molecular pathology in the context of the pathogenesis of non-neoplastic diseases, with a focus on the molecular processes involved in inflammation, repair and regeneration, immunity and control of normal and diseases tissue structure and organisation. The lectures will be delivered by a range of clinical and non-clinical academic staff, many of whom are internationally recognised experts in their field.
Aims
To equip students with an advanced knowledge of the molecular pathology and pathological processes underlying non-neoplastic diseases.
Intended Learning Outcomes
| Knowledge and Understanding |
On successful completion of the module students will have an understanding of the following areas of non-neoplastic molecular pathology as detailed below; Molecular Pathology Methods
Tissues and Organs
Regulation of Cell Differentiation and Growth
The Biology of Tissue Injury and Repair
The Immune Response and Immune-Mediated Disease
|
| Intellectual Skills |
|
| Transferable skills and personal qualities |
|
Teaching and Learning
The course contains 30 hours of lectures provided online (through the use of eLearning)
Assessment
SAQ examination (1 hour)
Written Assessment (2,000 words)
Feedback Methods
Students will be provided with mock papers before the examination and feedback will be provided within 15 working days. Written feedback will be provided within 15 working days.
Oncology Lecture Course MEDN 66620 (15 credits)
Course Unit Lead: David Cobben
The Tutorial unit is 15 credits (38 hours contact time, 112 hours learning time) and comprises two intensive one-week courses. A one-week course, covering the biological basis of chemotherapy, pharmacology and cancer biology, runs in November in the Auditorium of the Christie Hospital Education Centre. A second course covering the biological basis of radiotherapy, frontiers in cancer research and translational research, runs in February in the Ground Floor Seminar Room of the Wolfson Molecular Imaging Centre and in February in the Auditorium of the Christie Hospital Education Centre.
Course Unit Aim
To provide students with a detailed knowledge of the biological basis of cancer therapy
Intended Learning Outcomes
| Knowledge and understanding |
|
| Intellectual skills |
|
| Practical skills |
|
| Transferable skills and personal qualities |
|
Assessment
Two online SAQ (1 hour) 100%
Genomics of Common and Rare Inherited Diseases BIOL 67582 (15 Credits)
This unit aims to develop students’ knowledge of common and rare genetic disease. A comprehensive approach is taken to diagnosis, genomic testing, prognosis, management, inheritance and impact across a range of genetic conditions from common conditions, chromosome, single and heterogeneous conditions, inherited cancer, paediatric and adult onset conditions.
Limitations of practice, appropriate referral, psychosocial impact and family decision-making will equip students to work in multidisciplinary settings in the care for individuals and families with heritable conditions.
Learning will focus on a case-based approach to ensure relevance to clinical practice. Examples of paediatric and adult conditions will be used to illustrate the principles of genomic diagnosis, testing and counselling. Face to face sessions will be taught in one block over 3 days, to include lectures, problem based learning, and interactive workshops. This will be augmented by online lectures.
Course Unit Aims
The number of rare monogenic disorders is estimated to be greater than 7,000, but only in approximately half of these are the underlying genes known. Common diseases such as intellectual disability, diabetes, schizophrenia and autism are thought to arise from a complex interplay of genetic and environmental factors but deeper understanding of the genetic and mechanistic basis of these diseases is necessary for clinical translation.
The aim of this module is to provide a brief introduction to the clinical presentation and
manifestations of rare inherited and common diseases and consider the patient and family perspective with respect to the role and impact of genomics. The module will also focus on the genetic contribution to the aetiology of these conditions and strategies currently used to identify gene alterations in the clinical situation. Students will learn how to identify the most frequently encountered common and rare genetic disease phenotypes and how to select cases with unmet diagnostic need that will benefit from genomic testing.
Teaching and Learning Methods
Learning will focus on a case-based approach to ensure relevance to clinical practice. A range of formats will be used in both face-to-face and e-learning:
- Lectures, problem-based learning, case studies and group work
- eLearning: lectures, problem-based learning, case studies, and background reading to enhance face-to-face sessions
Assessment Methods
Written exam (50%)
Report (30%)
Oral assessment/presentation (20%)
Feedback Methods
Formative and Summative feedback given
Introduction to Immunology MEDN 67111 (15 Credits)
This module will provide students with an introduction to the area of immunology, providing a basic scientific knowledge and understanding of Immunology as a healthcare science discipline. Students will gain an appreciation of the role of clinical immunology and other blood science disciplines, in the investigation and management of disease, and will be equipped with the knowledge of principles of associated analytical techniques.
Aims
This module will provide the students with an introduction to the immune system and immune responses. They will understand the organisation and delivery of a clinical immunology laboratory service.
Teaching and Learning Methods
This unit employs a range of teaching and learning approaches including: lecturers, tutorials, case studies/presentations, problem-based learning and data analysis and interpretation exercises.
Intended Learning Outcomes
| Knowledge and Understanding |
|
| Intellectual skills |
|
| Practical skills |
|
| Transferable skills and personal qualities |
|
Assessment Methods
Written exam (50%)
Written assignment, inlcuidng essay (50%)
Feedback Methods
Marks will be provided online within 3/4 weeks of assessment deadline.
Principles of Haematology Oncology NURS 93160 (15 credits)
The course unit is designed to enable students to develop their knowledge and skills in relation to caring for the patient with a haematological malignancy.
The course unit is structured to enable qualified health professionals caring for individuals with a haematological cancer to remain in their own areas of practice to attain the course learning outcomes; although students have the option to negotiate access to service areas providing additional aspects of patient care in this context.
Course unit content follows three generic themes which follow the patient pathway; indications for treatment, the process of treatment and the range of treatment options and the effects of undergoing treatment for haematological malignancy, including rehabilitation, survivorship, supportive and palliative care and end of life care provision.
The course unit is constantly evolving in recognition of changing nature of clinical practice. This is undertaken through close collaboration between the University of Manchester and The Christie School of Oncology/Haematology department. This team approach aims to integrate theory into practice and is seen as an essential component of the course unit.
Course Unit Aims
- Develop student’s understanding of the pathophysiology of haematological malignancies and the aetiological factors, which may contribute to the development of haematological conditions/diseases within local, national and international populations.
- Facilitate students to critically reflect on current practice, identify areas for improvement and apply the evidence base to propose changes to patient care.
- Analyse the complex needs of haematology oncology patients within the context of changing healthcare provision and the use of evidence based practice.
- Analyse current national and local policy, Government and clinical guidelines, protocols for care delivery, evidenced based practice and their influence in the identification, treatment and management of haematological malignancy.
Intended Learning Outcomes
| Knowledge and Understanding |
|
| Intellectual skills |
|
| Practical skills |
|
| Transferable skills and personal qualities |
|
Assessment Methods
Written assignment, including essay (100%)
Feedback Methods
Students will normally have the opportunity to receive feedback on formative work submitted prior to the summative assessment. Other feedback opportunities will also be available in class and online discussion boards. Online feedback is provided in Grademark. Provisional feedback based on internal marking will be made available prior to the Exam Board on the basis that these marks are yet to be ratified at the Exam Board and therefore may be subject to change. A standard feedback mechanism in Grademark is utilised across all postgraduate programmes within the School which provides detailed and constructive feedback on each component and aspect of assessment and identifies areas of strength and those aspects which could be enhanced.
Student feedback is obtained through open discussion forums on blackboard, in class discussions, via formal University unit evaluation forms and also qualitative, in house evaluations at the end of the unit.
3. Teaching, Learning and Assessment
Postgraduate Taught Degree Regulations for Students
Students should familiarise themselves with the degree regulations for Postgraduate Taught Degrees by clicking on this link http://www.regulations.manchester.ac.uk/postgraduate-degree-regulations/ or reading the University document here: Introduction to the Postgraduate Degree Regulations for Students
Please note that students cannot progress to the MSc Paediatric Anaesthesia dissertation unless they have successfully completed 120 credits at Masters Level.
Guidance for Presentation of Taught Masters Dissertations
The University of Manchester guidance on presentation of taught Masters Dissertations is available at:
Guidance for the presentation of Taught Masters dissertations
The guidance explains the required presentation of the dissertation, and failure to follow the instructions in the guidance may result in the dissertation being rejected by the examiners.
Turnitin and Plagiarism
Plagiarism and Other Forms of Academic Malpractice
Academic malpractice is any activity - intentional or otherwise - that is likely to undermine the integrity essential to scholarship and research. It includes plagiarism, collusion, fabrication or falsification of results, and anything else that could result in unearned or undeserved credit for those committing it. Academic malpractice can result from a deliberate act of cheating or may be committed unintentionally. Whether intended or not, all incidents of academic malpractice will be treated seriously by the University.
The Faculty of Biology Medicine and Health have designed a learning module to raise your awareness of academic malpractice and how it can occur in general writing during your studies. This resource can be accessed via Blackboard - SMS Introductory Course and must be completed before you submit your first piece of academic writing for assessment.
The University provides workshops and online training via My Learning Essentials
Please refer to the University of Manchester guidance to students on plagiarism and other forms of academic malpractice
The full guidance document can be viewed here: http://documents.manchester.ac.uk/display.aspx?DocID=2870
Academic Malpractice: Procedure for the Handling of Cases can be found at: http://documents.manchester.ac.uk/DocuInfo.aspx?DocID=639
Turnitin
The University uses electronic systems for the purposes of detecting plagiarism and other forms of academic malpractice and for marking. Such systems include TurnitinUK, the plagiarism detection service used by the University.
As part of the formative and/or summative assessment process, you may be asked to submit electronic versions of your work to TurnitinUK and/or other electronic systems used by the University (this requirement may be in addition to a requirement to submit a paper copy of your work). If you are asked to do this, you must do so within the required timescales.
The School also reserves the right to submit work handed in by you for formative or summative assessment to TurnitinUK and/or other electronic systems used by the University.
Please note that when work is submitted to the relevant electronic systems, it may be copied and then stored in a database to allow appropriate checks to be made.
Mitigating Circumstances
Mitigating circumstances are personal or medical circumstances which are unforeseeable and unpreventable that could have a significant adverse effect on your academic performance. You should only submit a mitigating circumstances application if you consider it serious enough, and the timing critical, to have affected your performance in your assessed work and examinations.
Request for mitigation must be submitted to your programme administrator, in advance of your assessment submission deadline or exam. Requests for mitigation submitted after the assessment or exam (except those requests made as a result of circumstances that have arisen during the course of that assessment period) will not be considered without a credible and compelling explanation as to why the circumstances were not known before the beginning of the assessment period or why you were unable to complete or submit an application prior to the assessment or exam. Please note that not informing the University of circumstances due to personal feelings of embarrassment and pride, or having concerns over the confidential treatment of requests for mitigation, are not considered to be credible and compelling explanations
All mitigating circumstances applications must be supported by independent third party evidence. The type of evidence required will vary according to the nature of the circumstances. Examples of evidence include a doctor or other health professional’s letter, counsellor’s letter, self-certification form signed by your GP or GP’s Medical Practice (for illnesses of 7 days and under only). Please note that it is a University policy that the self-certification form must be signed by a GP; we cannot accept forms which have not been signed by a GP. Please note that if evidence has not been received within 2 weeks of the submission of your form, and you have not contacted them to inform them of any delay, your application will be refused and no further action will be taken.
Please ensure that you password protect or encrypt your mitigating circumstances form and supporting evidence before sending to your programme administrator.
Any requests for mitigation will be considered confidentially by a mitigating circumstances panel or sub-panel. Where a request for mitigation is supported, a recommendation will be made to the exam board for them to decide on the best course of action for the student.
You are advised to consult the following guidance, which directs you to seek advice and support before and whilst submitting a request for mitigation.
The University form and guidance for students is available on the web: A Basic Guide to Mitigating Circumstances.
For further information about the process and acceptable grounds for mitigation see: Mitigating Circumstances Policy & Procedures: http://documents.manchester.ac.uk/DocuInfo.aspx?DocID=4271
Please be advised that any requests need to be submitted by midday the day before the pre-arranged Mitigating Circumstances meeting.
23rd October 2019
27th November 2019
8th January 2020
19th February 2020
18th March 2020
22nd April 2020
20th May 2020
17th June 2020
Late Submission Penalty (Including Dissertation)
Work submitted after the deadline without prior approval will be subject to a late penalty in accordance with the University Policy on Submission of Work for Summative Assessment on Taught Programmes. The penalty applied is 10% of available marks deducted per day/24 hours (from the time of the original or extended deadline), until the assignment is submitted or no marks remain.
Penalties for late submission relate to 24 hours/calendar days, so include weekends and weekdays, as well as bank holidays and University closure days.
The mark awarded for the piece of work will be reduced by:
10% of the available marks deducted if up to 24 hours (1 day) late
20% of the available marks deducted if up to 48 hours (2 days) late
30% of the available marks deducted if up to 72 hours (3 days) late
40% of the available marks deducted if up to 96 hours (4 days) late
50% of the available marks deducted if up to 120 hours (5 days) late
60% of the available marks deducted if up to 144 hours (6 days) late
70% of the available marks deducted if up to 168 hours (7 days) late
80% of the available marks deducted if up to 192 hours (8 days) late
90% of the available marks deducted if up to 216 hours (9 days) late
100% of the available marks deducted if up to 240 hours (10 days) late
If the assessment is submitted within 10 days of the deadline the assessment should be marked and feedback to the student provided. If this mark before the penalty is applied reaches the appropriate pass mark but the applied penalty results in a fail of the assessment, the student should not be required to resit the assessment as the original mark can be taken as the resit mark. Further information and examples can be found in the Policy and associated Guidance documents.
For work submitted more than 10 days late, it is regarded as a non-submission and need not be marked. In this case a mark of zero will be awarded and normal resit regulations will apply.
The sliding scale should only be applied to first-sit submissions. For all referred (resit) assessment, any late submission will automatically receive a mark of zero.
For further information:
Policy on the Submission of Work for Summative Assessment on Taught Programmes
Assignment Word Count (Including Dissertation)
In accordance with the University Policy on Marking:
Each written assignment has a word limit which you must state at the top of your first page. It is acceptable, without penalty, for you to submit an assignment within a range that is plus 10% of this limit. If you present an assignment with a word count exceeding the specified limit+10%, the assignment will be marked but 1% will be deducted from this mark for every 100 words over the limit given.
For an original word limit that is 1000 words and an assignment that is marked out of 100. If a submission is made that is 1101 words then it exceeded the 10% leeway, and is more than 100 words over the original limit and should receive a 1 mark deduction.
In accordance with accepted academic practice, when submitting any written assignment for summative assessment, the notion of a word count includes the following without exception:
- All titles or headings that form part of the actual text. This does not include the fly page or reference list
- All words that form the actual essay
- All words forming the titles for figures, tables and boxes, are included but this does not include boxes or tables or figures themselves
- All in-text (that is bracketed) references
- All directly quoted material
Certain assessments may require different penalties for word limits to be applied. For example, if part of the requirement for the assessment is conciseness of presentation of facts and arguments. In such cases it may be that no 10% leeway is allowed and penalties applied may be stricter than described above. In such cases the rules for word count limits and the penalties to be applied will be clearly stated in the assessment brief and in the submission details for that assessment.
Fitness to Practise
Where a programme of study requires the student to undertake practical training in a quasi-professional role in relation to patients, clients or service-users or where the qualification provides a direct license to practise, the Faculty of Biology, Medicine and Health has a duty to ensure that the student is fit to practise. In order to protect present or future patients, clients or service users and to comply with the requirements of professional/regulatory bodies, the Faculty of Biology, Medicine and Health has established a procedure for dealing with student-related fitness to practise issues.
Fitness to Practise issues are initially investigated and considered locally within the School (e.g. by a Health and Conduct Committee) and if necessary referred to the Faculty Fitness to Practise Committee.
A student may appeal against the decision of a Fitness to Practise Committee within twenty days of the decision but only on one or more of the following grounds:
a) procedural irregularity;
b) availability of new evidence which could not reasonably have been expected to be presented to the original hearing;
c) the disproportionate nature of the penalty.
The TLSO facilitates the arrangements for Fitness to Practise Appeals Committees. An Appeals Committee has the power to confirm or alter the original decision, and the outcome is confirmed to students in a Completion of Procedures letter. A student may then decide to pursue a complaint with the OIA.
Information on Fitness to Practise related matters can be found at: www.tlso.manchester.ac.uk/appeals-complaints/fitnesstopractise
Academic Appeals, Complaints, Conduct and Discipline
Academic Appeals
- Students have a right of appeal against a final decision of an Examination Board, or a progress committee, or a graduate committee or equivalent body which affects their academic status or progress in the University.
- Students thinking of appealing should first discuss the matter informally with an appropriate member of staff, in order to better understand the reason for the result or decision.
- Should you wish to proceed to a formal appeal, this must be submitted within the timeframe outlined in the Academic Appeals Procedure to the Faculty Appeals and Complaints Team, Room 3.21, Simon Building, University of Manchester, M13 9PL (e-mail: FBMHappealsandcomplaints@manchester.ac.uk).
- The Academic Appeals Procedure (Regulation XIX) and associated documents, including the form on which formal appeals should be submitted, can be found at www.regulations.manchester.ac.uk/academic
Student Complaints
- The University’s Student Complaints Procedure (Regulation XVIII) and associated documents, including a complaints form, can be found at www.regulations.manchester.ac.uk/academic
- The University has separate procedures to address complaints of bullying, harassment, discrimination and/or victimisation - see https://www.reportandsupport.manchester.ac.uk/
- Students thinking of submitting a formal complaint should, in most instances, attempt informal resolution first (see the procedure). Formal complaints should be submitted on the relevant form to Faculty Appeals and Complaints Team, Room 3.21, Simon Building, University of Manchester, M13 9PL (e-mail: FBMHappealsandcomplaints@manchester.ac.uk).
Conduct and Discipline of Students
- General University information on the conduct and discipline of students can be found at www.tlso.manchester.ac.uk/appeals-complaints/conductanddisciplineofstudents/
- Faculty policies for students on communication and dress code, social networking. and drugs and alcohol can be found at:
- http://documents.manchester.ac.uk/display.aspx?DocID=29038 (Communication and Dress Code)
- http://documents.manchester.ac.uk/display.aspx?DocID=29039 (Drugs and Alcohol)
- http://documents.manchester.ac.uk/display.aspx?DocID=29040 (Social Networking)
- Information on Academic Malpractice and how to avoid it can be found at http://www.regulations.manchester.ac.uk/guidance-to-students-on-plagiarism-and-other-forms-of-academic-malpractice/
- In accordance with the Policy on Submission of Work for Summative Assessment on Taught Programmes, ‘All typed summative assessment, including dissertations, should be submitted online and subjected to plagiarism detection software, where appropriate’.
4. Student Progression
Monitoring Attendance and Wellbeing of Students
The programme director and teaching staff will monitor the work and attendance of students on the programme. This is for your benefit and helps to ensure you are coping with the work. Regular or a pattern of non-attendance and/or engagement will result in you being contacted by the School to meet with your programme director. Following this, further action will be taken if there isn’t a significant improvement in attendance.
For further information see:
Regulation XX Monitoring Attendance and Wellbeing of Students
The University offers a range of advice and support to students experiencing problems with attendance. The A-Z of Services can be found on the MyManchester website. Here you can find a information on a wide range of topics such as library services, disability support and careers advice.
You can also speak to your Programme Director and/or Academic Advisor.
What to do if you are absent
In case of illness you should supply a doctor’s certificate or, if the illness is brief, a self-certification. If you are absent for other reasons then you should write a letter to the Programme Director explaining the circumstances. Medical certificates or letters should be given in person or sent to the Programme Administrator. Whatever your reason for being away, tell your supervisor about it and make any necessary arrangements to catch up with work you have missed.
Special Permissions
Interruptions to programme and extensions to writing up
It is the expectation of the University that postgraduate taught students pursue their studies on a continuous basis for the stipulated duration of their programme. However, it is recognised that students may encounter personal difficulties or situations which may seriously disrupt or delay their studies. In some cases, an interruption or extension to your programme of study may be the most sensible option.
Students who wish to interrupt the programme or extend to write up the dissertation should initially discuss their plans and reasons with the Programme Director and/or their Academic Advisor.
Students should also provide documentary evidence when appropriate, for example, doctor’s letter, sick note etc.
The forms required for formal application are available from your Programme Administrator.
Tier 4 Visa Attendance Monitoring Census
The University operates attendance monitoring census points within the academic year in order to confirm the attendance of students holding a Tier 4 Student Visa. This is to ensure the University meets the UKVI statutory requirements as a sponsor of Tier 4 students and its responsibilities in accordance with its Highly Trusted Sponsor status.
If you are a Tier 4 visa holder, you must attend these attendance monitoring census points, in addition to complying with your programme’s attendance requirements.
When are the census points?
In the 2019/20 academic year, the attendance monitoring census points will be during the following periods:
- 23rd September - 4th October 2019
- 13th January - 24th January 2020
- 13th May - 3rd June 2020
- 13th July - 24th July 2020
Please note:
- If you are a new student, registration is your first point to confirm your attendance at the University and you will not be required to attend a separate census point in September/October 2019.
- You will receive an e-mail from your programme administrator to confirm when and where you should go to have your attendance confirmed. You must check your University e-mail account regularly. Failure to check your e-mail account is not a valid reason to be absent from a census point.
What if a Tier 4 student cannot attend a census point?
If you cannot attend in person due to a valid reason which includes: illness; placement; field studies; on year abroad; research work; or any other reason connected to your programme of study, you must email your programme administrator to inform us of your absence and your inability to attend in person. In the case of illness, you must provide a copy of a medical certificate. If you are in this position you should report in person to the School as soon as possible after you return to campus.
Students who are recorded as interrupting their studies are not expected to attend during their period of interruption.
What happens if a student does not attend a census point?
The School must be able to confirm your presence to the UKVI by the end of each census point in the academic year. If you do not attend a census point when required by your School and you do not provide a valid explanation for your absence you will be deemed to be “not in attendance”.
Those students identified as “not in attendance” will be reported to the UKVI and the University will cease to sponsor the student’s Tier 4 visa. The Tier 4 visa will then be curtailed and the student must leave the UK within 60 days.
Further information
For more information on Tier 4 visas: https://www.gov.uk/tier-4-general-visa
If you have any concerns about the attendance monitoring census points, or your Tier 4 visa status, please contact visa@manchester.ac.uk
Withdrawal from the Programme
Students who are considering withdrawing from the programme should discuss this in the first instance with the Programme Director.
If arrangements for withdrawal need to be made, this will be handled by the Programme Administrator, who will manage communication with the Fees and Records Departments and other University bodies as appropriate OR Students may liaise directly with the Programme Administrator who will communicate this information directly to the University Student Services Centre.
5. Support and Guidance
Student Support and Guidance
Student support and guidance within the programme
Support and advice is available to all students both formally and informally from the Programme Directors, the Programme Administrator and research project supervisors.
If you have any queries or would like to discuss any issues at all – academic, administrative, technical or personal – please do not hesitate to get in touch. All personal issues will be dealt with confidentially.
If we are unable to help you directly, we can put you in touch with many of the support services that are available to students of the University through our Student Services Centre. You can approach these services independently, without the involvement of programme staff. Use the A-Z of Student Services Guide as an additional source of information.
Student support for the dissertation
During the research project and writing up of the dissertation, students will have individual support from their research project supervisor and scheduled structured sessions to monitor their progress and provide support, with help being offered if any problems are being encountered. In addition, students are made aware that they have the option of contacting the programme directors at any time if they are experiencing difficulties, whether this is in relation to their project, or indeed, with regard to any other issue of relevance.
Student support from the University
The University offers a range of support and guidance services to students, for example, Student Health Service, Student Union Advice Centre, Student Counselling and Careers Advice. Details of all these services can be obtained from the A-Z of Student Services
Counselling Service
The Counselling Service is available for all students. It is free and consists of a team of professional counsellors. The service provides confidential counselling for anyone who wants help with personal problems affecting their work or well-being.
The service is open 9.00am to 5.00pm Monday to Friday all year round except public holidays.
Occupational Health
The Occupational Health service is a specialised area of medicine concerned with the way in which an individual’s health can affect his or her ability to do a job and to study and conversely how the work environment can affect an individual’s health. Their aim is to promote the physical, mental and social well-being of students and to reduce the incidence of ill-health arising from exposure to work place hazards.
Students Union Advice Centre
The Students Union has advisers who can help with any matter ranging from finances to housing and beyond. On the South Campus, the Advice Centre is on the first floor in the Student Union Building, and is open Monday to Friday, 10.00am to 4.00pm, term time and vacation. There is no need to make an appointment.
Academic Success Programme
You’re studying at the University of Manchester – congratulations! Writing and speaking Academic English can be challenging, even for native speakers. Our team of experienced tutors are here to support you, and will help boost your confidence to work independently in English through a series of interactive workshops - freely available to all University of Manchester students.
To find out more, and to register, please go to www.manchester.ac.uk/academicsuccessprogramme
Disability Advisory and Support Service (DASS)
The University of Manchester welcomes students with a disability or specific learning difficulties. The University has a Disability Advisory and Support Service, who can supply further information and DASS advisors will be pleased to meet you to discuss you needs. DASS will liaise with your School through the Disability Coordinator to make the necessary arrangements for your support during your time in Manchester.
The DASS office can also provide a copy of the University’s Disability Statement, ‘Opportunities for Students with Additional Support Needs at the University of Manchester’ which sets out the policy and provision for students with a disability.
DASS Contact Details:-
Location: 2nd Floor, University Place
Email: dso@manchester.ac.uk; Phone: 0161 275 7512
Text: 07899 658 790; Website: www.dso.manchester.ac.uk/
Disability Coordinator Contact Details:-
Name: Kay Gardner (0161 306 7978) & Wiktoria Obidniak-Marciniak (0161 306 5519)
Email: kay.gardner@manchester.ac.uk or wiktoria.obidniak-marciniak@manchester.ac.uk
Religious Observance and Looking after yourself and your patients during Ramadan
Policy on Religious Observance:
- University Policy
- The Faculty of Biology, Medicine and Health has produced guidance for healthcare students on fasting and caring: Fasting and Caring - Looking after yourself and your patients during Ramadan: guidance for health care students.
6. Student Representation and Feedback
Student Representation and Feedback
Students, in consultation with the Programme Administrator, should arrange an informal election of their Student Representative near the beginning of the academic year. If more than one person is interested in the role, then each candidate is asked to write a short proposal which is circulated to the student body, and an election is held.
The overall responsibilities of the Student Representative are
- to liaise between staff and students about matters of concern
- to provide two-way feedback on programme and teaching quality
- to promote active student involvement in the development of the programme
- to identify student issues and needs on the programme
- to attend programme committee meetings representing the student voice
- to find effective ways to feedback the outcomes of meetings to the student body
- to attend relevant student representative training
- to liaise with other Student Representatives to gain support and ideas
- to become established as a central point for information and guidance for students in the group.
Student representatives are not required to get involved with fellow students’ personal problems, academic difficulties, or individual student allegations of unfair or inappropriate treatment.
Confidentiality is imperative when dealing with student issues. The representative is chosen by fellow students and has their trust, and must maintain it. Any discussion of an individual student’s situation with a third party requires their consent beforehand.
7. Programme Management
Programme Management and Committee Structure
Programme Management
The programme is managed and operated in accordance with the policies, principles, regulations and procedures of the University of Manchester.
Programme Directors relate to the School and Faculty Postgraduate Teaching Committees on matters relating to admissions, exams, reviews and approval of new programmes and units, quality assurance etc. and policy issues of broad relevance to the Graduate School.
The Programme Committee will meet each semester and consist of the Programme Director, Programme Administrator, Programme Committee members and the unit co-ordinators.
The remit of the committee will be to:
- Oversee the teaching, assessment and examining arrangements;
- Monitor cohort progression including failure rate, withdrawal rate;
- Evaluate the extent to which the learning outcomes are achieved by students;
- Monitor, maintain and enhance standards of all aspects of the programme;
- Evaluate the effectiveness of the curriculum and of assessment in relation to programme learning outcomes;
- Evaluate the effectiveness and relevance of the teaching and learning methods employed;
- Review and revise the programme in the light of any relevant Quality Assurance Agency (QAA) benchmarks, any other relevant external and/or professional requirements and developing knowledge in the subject area;
- Receive, consider and respond to feedback from students, employers and external examiners;
- Where the need for change is identified, effect the changes quickly and efficiently;
- Produce an annual action plan via annual monitoring;
- Produce reports for periodic review
- Produce relevant information for an Institutional Audit;
- Review programme documentation, e.g., programme handbooks, programme specifications, promotional literature and programme website;
- Ensure suitable and efficient arrangements are in place for recruitment, admission and induction.
Committee Structure
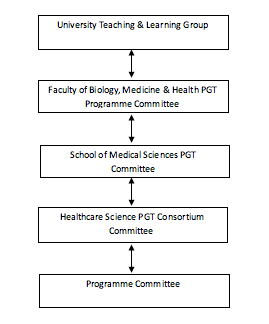
The Programme Committee acts as a curriculum development team for the Programme. The Programme Committee will report to a School, or Department, or Faculty level committee. The Programme Director is responsible for the management of the programme, and the Programme Committee is established to support the Programme Director in the carrying out of their responsibilities.
The role of the External Examiner
External Examiners are individuals from another institution or organisation who monitor the assessment processes of the University to ensure fairness and academic standards. They ensure that assessment and examination procedures have been fairly and properly implemented and that decisions have been made after appropriate deliberation. They also ensure that standards of awards and levels of student performance are at least comparable with those in equivalent higher education institutions.
External Examiners’ reports
External Examiners’ reports relating to this programme will be shared with student representatives and details of any actions carried out by the programme team/School in response to the External Examiners’ comments will be discussed. Students should contact their student representatives if they require any further information about External Examiners’ reports or the process for considering them.”
External Examiner Details
The External Examiner for this programme is Professor Uta Griesenback, Professor of Molecular Medicine at the National Heart and Lung Institute, Imperial College London
Please note that it is inappropriate for students to make direct contact with External Examiners under any circumstances, in particular with regards to a student’s individual performance in assessments. Other appropriate mechanisms are available for students, including the University’s appeals or complaints procedures and the UMSU Advice Centre. In cases where a student does contact an External Examiner directly, External Examiners have been requested not to respond to direct queries. Instead, External Examiners should report the matter to their School contact who will then contact the student to remind them of the other methods available for students. If students have any queries concerning this, they should contact their Programme Office (or equivalent).
8. Learning Resources
Learning Resources
Libraries
All registered students may become members of the University of Manchester Library on the main campus.
Up-to-date news about the library is available here.
IT Services and eLearning
IT Services Support Centre online
Details of what IT support is available and how to access it can be found on the FBMH eLearning Support page.
Login to the Support Centre online to log a request, book an appointment for an IT visit, or search the Knowledge Base.
Telephone: +44 (0)161 306 5544 (or extension 65544). Telephone support is available 24 hours a day, seven days a week.
In person: Walk-up help and support is available at the Joule Library, Main Library or Alan Gilbert Learning Commons:
Use Support Centre online for support with eLearning, from where you may make a request, report a fault, or search the Knowledge Base. The email address is: elearning@manchester.ac.uk
Blackboard
Blackboard, the University's 'virtual learning environment', will be used for online teaching.
What is Blackboard?
Blackboard is a web-based system that complements and builds upon traditional learning methods used at The University of Manchester. By using Blackboard you can
- view course materials and learning resources,
- communicate with lectures and other students,
- collaborate in groups,
- get feedback
- submit assignments
- monitoring your own progress at a time and place of your own convenience.
Training in the use of software
The Faculty eLearning team have produced a short introduction to Blackboard for new students. The recording is hosted in two places: the VLS and on YouTube:
The recording is just over seven minutes long and covers most of the commonly used tools in Blackboard.
9. Useful Links
Academic and Student Support Policies
Academic Support Policies
A full list of University Policies and documents
Academic Appeals (Regulation XIX)
Academic Malpractice: Procedure for the Handling of Cases
Basic Guide to Student Complaints
Conduct and Discipline of Students (Regulations XVII)
General University information on the Conduct and Discipline of Students
Faculty policies for students on Communication and Dress Code, Social Networking and Drugs & Alcohol can be found at:
Information on Academic Malpractice and how to avoid it
Guidance for the Presentation of Taught Masters Dissertations
Guidance to Students on Plagiarism and Other Forms of Academic Malpractice
Policy on Submission of Work for Summative Assessment on Taught Programmes
Policy on Mitigating Circumstances
Mitigating Circumstances Guidance for Students
Policy on Feedback to Undergraduate and Postgraduate Taught Students
Work and Attendance of Students (Regulation XX)
Blackboard
Students should access Blackboard via My Manchester
Disability Advisory and Support Service
University Language Centre – Study English - Tel: 0161 306 3397
Equality, Diversity and Inclusion for Staff and Students
Occupational Health Services for Students
A Personal Safety Guide for International Students
10. Appendix
Roles and Responsibilities of Supervisors and Students
Roles and Responsibilities of Supervisors
The responsibilities of Supervisors include:
a) giving guidance about the nature of research and the standard expected, the planning of the research programme, literature and sources, attendance at taught classes where, appropriate and about requisite techniques (including arranging for instruction where necessary);
b) maintaining contact through regular meetings (the frequency of meetings being appropriate to the research being undertaken and agreed in advance);
c) being accessible to the student at other appropriate times for advice and responding to difficulties raised by the student;
d) giving detailed advice on the necessary completion dates of successive stages of the work so that the thesis may be submitted within the agreed timescale;
e) requesting written work or reports as appropriate and returning written material with constructive criticism and in reasonable time;
f) ensuring that for degrees where an oral examination is required the student is adequately prepared by arranging for the student to present his or her work to staff and graduate seminars;
g) ensuring that the student is made aware when progress is not satisfactory and facilitating improvement with advice and guidance;
h) establishing at an early stage the Supervisor's responsibilities in relation to the student's written work, including the nature of the guidance and comments to be offered as the work proceeds and on the draft of the thesis before it is submitted. It must be made clear to the student that research for a higher degree is undertaken within the general principle that a thesis must be the student's own work;
i) ensuring that at the end of each year of the course the student produces a research report, to which the Supervisor should add comments on progress. The Supervisor's comments on progress should be signed by the student to confirm that they have been seen, before the annotated report is submitted by the Supervisor to the appropriate Supervisory body in accordance with established Graduate School procedures;
j) making students aware of other researchers and research work in the department and Graduate School;
k) encouraging the student to publish the research;
l) providing pastoral support and advising students, where appropriate, of University support services;
m) bringing to the attention of the students the health and safety regulations and academic rules, regulations and codes of practice of the University. More detailed guidance on Health and Safety is available in the University's Health and Safety Policy Notice UMHSP 33, available from Health and Safety Services, which interprets and applies the CVCP Note of Guidance N/93/111, "Health and Safety Responsibilities of Supervisors towards Graduate and Undergraduate students". Guidance on specific situations is available from the staff of Health and Safety Services;
n) to recommend examiners for the student’s thesis after discussion with the student to ensure that the proposed examiners have not had a significant input into the project, a significant personal, financial or professional relationship with the student, or that there is no other good reason to doubt the suitability of the recommendation.
Responsibilities of the Student
The responsibilities of the student include:
a) pursuing the programme with a positive commitment, taking full advantage of the resources and facilities offered by the academic environment and, in particular, contact with the Supervisor, other staff and research students;
b) discussing with the Supervisor the type of guidance and comment believed to be most helpful, and agreeing a schedule of meetings;
c) ensuring that he/she is aware of the health and safety regulations and academic rules and regulations and codes of practice of the University;
d) successfully completing any training programme arranged within the prescribed time period;
e) taking the initiative in raising problems or difficulties, however elementary they may seem, bearing in mind that prompt discussion and resolution of problems can prevent difficulties and disagreements at a later stage;
f) maintaining the progress of the work in accordance with the stages agreed with the Supervisor, including in particular the presentation of written material as required, in sufficient time to allow for comments and discussion before proceeding to the next stage. Where possible, students will be given details of the work programme for the academic year at the beginning of the year;
g) providing at the end of each year a report on progress to the Supervisor for submission to the appropriate Supervisory body in accordance with established Graduate School procedures. The report at the end of the first year will normally be a substantial piece of work in accordance with Graduate School procedures. Reports at the ends of subsequent years, occurring before the completion of the thesis, should adequately describe the progress made during the preceding year. If industrial research contract reports are being written on a regular basis, the Supervisor might judge these to be a satisfactory alternative to progress reports;
h) agreeing with the Supervisor the amount of time to be devoted to the research and the timing and duration of holiday periods;
i) deciding when to submit the thesis. The student should take due account of the Supervisor's opinion but must recognise that it is only Advisory. The student must ensure that appropriate notice of intent to submit a thesis is given, in accordance with the published University procedures;
j) checking the completeness and accuracy of the text of the thesis submitted; failure to check the thesis carefully may result in the thesis being failed or cause a delay in the award of a degree;
k) to disclose, in discussion with Supervisors concerning potential examiners for the thesis, any information that could significantly affect the suitability of the proposed examiner (s). Such information may concern a significant input from the examiner(s) into the project or a significant personal, financial or professional relationship they may have had, with the student.
